Water as a shape-shifting and active biological building block
Why study water structure and dynamics near active surfaces?
The design of surfaces with tuned activity for adsorption and interaction in water is of enormous interest to de novo protein design, bioengineering of active protein surfaces, mimicking ice binding proteins, design polymer-based soft matter surfaces with tuned activity or the rational design of catalyst supports with optimal transport, adsorption, and stability. The Han Lab has developed and disseminated a quantum sensing approach known as Overhauser DNP that relies on double-quantum flip-flop transition between a dipolar coupled electron spin of a spin label and a 1H nuclear spin of water to selectively probe equilibrium water diffusion near surfaces by utilizing the dependence of the electron-nuclear spin coupling on the motional speed of water relative to the spin label tethered to the surface of interest.
Knowledge Gap?
The molecular origin of biological surface activity is poorly understood. In particular, the role of surface water dynamics, structure and thermodynamics have been elusive to experimental characterization for complex systems. Current literature does not offer a clear and rational explanation for the link between water structure and dynamics, and few techniques offer surface sensitivity or nanometer scale spatial resolution to directly measure equilibrium water properties. The questions that the Han lab is pursuing include the design of active sites and active surfaces, the encoding pinning sites to the protein surface to program directed assembly, and the role of water in transforming light energy to mechanical energy by hydraulic action.

Unique Han Lab Tools and Concepts?
The Han lab has developed and uses state of the art magnetic resonance tools (e.g., ODNP and EPR) to generate unambiguous 1H signatures of water distinct from signatures of the biomolecular surface, as well as the 17O NMR chemical shift and quadrupolar coupling constant of water that is exceptionally sensitive to even the slightest perturbation to the local hydrogen bond and electronic density of involved water. Its core strategy is two-pronged: the direct experimental measurements of site-specific water dynamics via ODNP near model protein and polymer surfaces and the direct experimental measurements of water structure via 17O NMR chemical shift that is a direct probe of 17O electronic structure, coupled with electronic structure calculations of water clusters.
Future Work?
Future studies will focus on shaping the water structure and the activity of biomolecular and soft matter surfaces. We aim to understand the molecular basis of surface activity of biomolecular and soft matter structures, with an emphasis on resolving the structure and thermodynamics of organized water hydrating soft, dynamic, surfaces. This ambitious goal has been elusive to solve due to lack of measurement technology—this is the gap that the Han lab is pursuing to close

Featured Publications
- Computation of Overhauser Dynamic Nuclear Polarization processes reveals fundamental correlation between water dynamics, structure, and solvent restructuring entropy
-
Bridging the Gap in Cryopreservation Mechanism: Unraveling the Interplay between Structure, Dynamics, and Thermodynamics in Cryoprotectant Aqueous Solutions
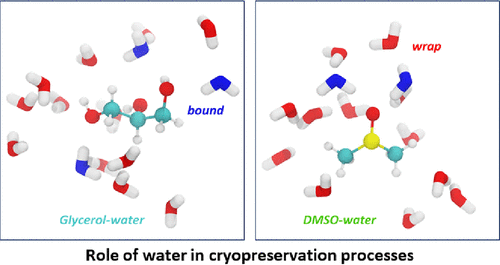
Cryoprotectants play a crucial role in preserving biological material, ensuring their viability during storage and facilitating crucial applications such as the conservation of medical compounds, tissues, and organs for transplantation. However, the precise mechanism by which cryoprotectants modulate the thermodynamic properties of water to impede the formation and growth of ice crystals, thus preventing long-term damage, remains elusive. This is evident in the use of empirically optimized recipes for mixtures that typically contain DMSO, glycerol, and various sugar constituents. Here, we use terahertz calorimetry, Overhauser nuclear polarization, and molecular dynamics simulations to show that DMSO exhibits a robust structuring effect on water around its methyl groups, reaching a maximum at a DMSO mole fraction of XDMSO = 0.33. In contrast, glycerol exerts a smaller water-structuring effect, even at higher concentrations (Scheme 1). These results potentially suggest that the wrapped water around DMSO’s methyl group, which can be evicted upon ligand binding, may render DMSO a more surface-active cryoprotectant than glycerol, while glycerol may participate more as a viscogen that acts on the entire sample. These findings shed light on the molecular intricacies of cryoprotectant solvation behavior and have potentially significant implications for optimizing cryopreservation protocols.
