Online Publications

Google Scholar Publication List
View all publications
Pubmed Publication List
View all publications2026 Journal Articles
Exchange coupling-assisted 13C dynamic nuclear polarization in microdiamond at 14 T
Q. Stern, J. Cui, R. Chaklashiya, C. Tobar, M. Judd, O. Nir-Arad, D. Shimon, I. Kaminker, H. Takahashi, J. R. Sirigiri, and S. Han Phys. Chem. Chem. Phys., 2026. https://doi.org/10.1039/D5CP04594K
We investigate nitrogen substitution defects, also known as P1 centers, in Type 1b diamonds generated under high pressure and high temperature (HPHT) as a source of electron spin polarization. The open question was how readily electron spin polarization in this diamond transfers to 13C nuclear spins within the diamond matrix at 14 T by dynamic nuclear polarization (DNP). The goal was to refine the model for clustered P1 centers in HPHT diamonds and evaluate their potential as a source for DNP hyperpolarization or contrast. The study relied on frequency-stepped measurements of DNP profiles under magic angle spinning (MAS) using the mm-wave output of a frequency-tunable gyrotron and a regular superconducting NMR magnet at a single field. We observe up to 700-fold 13C on/off signal enhancements in both MAS and static mode at room temperature, and 130-fold between 35 and 100 K. Modelling the experimental results revealed the dominant role of P1 clusters harboring inter-P1 dipolar and exchange couplings exceeding 100 MHz in achieving 13C DNP at 14.1 T. Our results exemplify the importance of exchange coupling for high-field DNP and provides a refined model for DNP via P1 centers of HPHT diamonds.
Exchange coupling-assisted 13C dynamic nuclear polarization in microdiamond at 14 T
Q. Stern, J. Cui, R. Chaklashiya, C. Tobar, M. Judd, O. Nir-Arad, D. Shimon, I. Kaminker, H. Takahashi, J. R. Sirigiri, and S. Han Phys. Chem. Chem. Phys., 2026. https://doi.org/10.1039/D5CP04594K
We investigate nitrogen substitution defects, also known as P1 centers, in Type 1b diamonds generated under high pressure and high temperature (HPHT) as a source of electron spin polarization. The open question was how readily electron spin polarization in this diamond transfers to 13C nuclear spins within the diamond matrix at 14 T by dynamic nuclear polarization (DNP). The goal was to refine the model for clustered P1 centers in HPHT diamonds and evaluate their potential as a source for DNP hyperpolarization or contrast. The study relied on frequency-stepped measurements of DNP profiles under magic angle spinning (MAS) using the mm-wave output of a frequency-tunable gyrotron and a regular superconducting NMR magnet at a single field. We observe up to 700-fold 13C on/off signal enhancements in both MAS and static mode at room temperature, and 130-fold between 35 and 100 K. Modelling the experimental results revealed the dominant role of P1 clusters harboring inter-P1 dipolar and exchange couplings exceeding 100 MHz in achieving 13C DNP at 14.1 T. Our results exemplify the importance of exchange coupling for high-field DNP and provides a refined model for DNP via P1 centers of HPHT diamonds.
2025 Journal Articles
Coherent Control over Nuclear Hyperpolarization Using an Optically Initializable Chromophore-Radical System
Chromophore radicals (CR) are emerging as important components for molecular quantum information science (QIS), especially in the context of quantum sensing. Here, we demonstrate that the optically hyperpolarized electrons in a 1,6,7,12-tetrakis(4-tert-butylphenoxy)-perylene-3,4,9,10-bis(dicarboximide) (tpPDI) covalently linked to a partially deuterated 1,3-bis(diphenylene)-d16-2-phenylallyl radical (BDPA-d16) can be coherently manipulated via pulsed dynamic nuclear polarization (DNP) methods to transfer polarization to nuclear spins and back. Under light illumination at 85 K, electron hyperpolarization in BDPA is enhanced 2.1- to 2.4-fold over thermal polarization and lasts for more than 100 ms. By applying nuclear orientation via electron spin-locking (NOVEL) DNP, this optically amplified electron hyperpolarization was successfully transferred to a 1H nuclear spin within the CR system and efficiently returned to the electron spin for readout via reverse-NOVEL. The NOVEL transfer efficiency of 65% amounts to a 688-fold nuclear spin hyperpolarization of the target nuclear spin, considering the 2.1-fold electron spin hyperpolarization. This reversible coherent manipulation of hyperpolarization transfer highlights the utility of CR systems to initialize and read out nuclear spin states in a disordered matrix at moderate cryogenic temperatures. Coupled with CRs’ environmental compatibility, tunability, and precise state initialization, these results highlight the promising role of nuclear spins in CRs for QIS applications, including quantum sensing and memory.
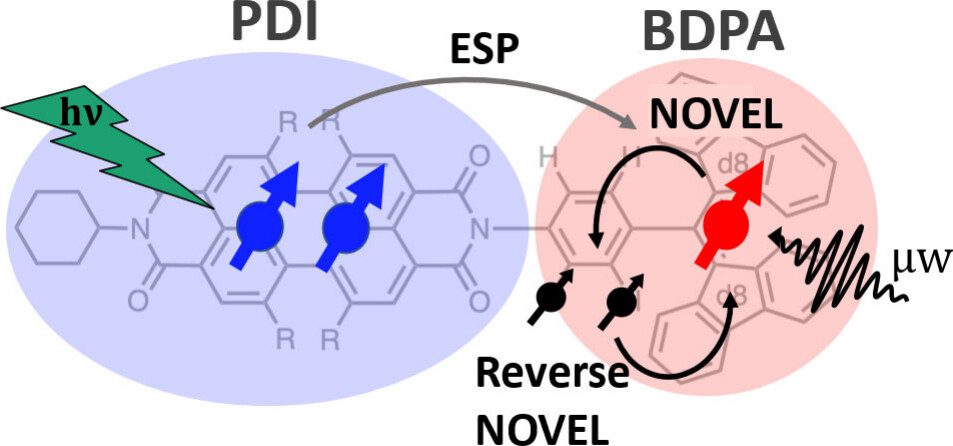
Coherent Control over Nuclear Hyperpolarization Using an Optically Initializable Chromophore-Radical System
Chromophore radicals (CR) are emerging as important components for molecular quantum information science (QIS), especially in the context of quantum sensing. Here, we demonstrate that the optically hyperpolarized electrons in a 1,6,7,12-tetrakis(4-tert-butylphenoxy)-perylene-3,4,9,10-bis(dicarboximide) (tpPDI) covalently linked to a partially deuterated 1,3-bis(diphenylene)-d16-2-phenylallyl radical (BDPA-d16) can be coherently manipulated via pulsed dynamic nuclear polarization (DNP) methods to transfer polarization to nuclear spins and back. Under light illumination at 85 K, electron hyperpolarization in BDPA is enhanced 2.1- to 2.4-fold over thermal polarization and lasts for more than 100 ms. By applying nuclear orientation via electron spin-locking (NOVEL) DNP, this optically amplified electron hyperpolarization was successfully transferred to a 1H nuclear spin within the CR system and efficiently returned to the electron spin for readout via reverse-NOVEL. The NOVEL transfer efficiency of 65% amounts to a 688-fold nuclear spin hyperpolarization of the target nuclear spin, considering the 2.1-fold electron spin hyperpolarization. This reversible coherent manipulation of hyperpolarization transfer highlights the utility of CR systems to initialize and read out nuclear spin states in a disordered matrix at moderate cryogenic temperatures. Coupled with CRs’ environmental compatibility, tunability, and precise state initialization, these results highlight the promising role of nuclear spins in CRs for QIS applications, including quantum sensing and memory.

Structure-specific Mini-Prion Model for Alzheimer’s Disease Tau Fibrils
A critical discovery of the past decade is that tau protein fibrils adopt disease-specific hallmark structures in each tauopathy. The faithful generation of synthetic fibrils adopting hallmark structures that can serve as targets for developing diagnostic and/or therapeutic strategies remains a grand challenge. We report on a rational design of synthetic fibrils built of a short peptide that adopts a critical structural motif in tauopathy fibrils found in Alzheimer’s Disease (AD) and Chronic Traumatic Encephalopathy (CTE). They serve as minimal prions with exquisite seeding competency, in vitro and in tau biosensor cells, for recruiting tau constructs ten times larger its size en route to AD or CTE fibril structures. We demonstrate that the generation of AD and CTE-like fibril structures is dramatically catalyzed in the presence of mini-AD prions and further influenced by salt composition in solution. Double Electron-Electron Resonance studies confirmed the preservation of AD-like folds across multi-generational seeding. Fibrils formed with the full AD/CTE-like core show strong seeding competency, with their templating effect dominating over the choice of salt composition that tunes the initial selection of AD- and CTE-like fibril populations. The mini-AD prions serve as a potent catalyst with templating capabilities that offer a novel strategy to design pathological tau fibril models.
Structure-specific Mini-Prion Model for Alzheimer’s Disease Tau Fibrils
A critical discovery of the past decade is that tau protein fibrils adopt disease-specific hallmark structures in each tauopathy. The faithful generation of synthetic fibrils adopting hallmark structures that can serve as targets for developing diagnostic and/or therapeutic strategies remains a grand challenge. We report on a rational design of synthetic fibrils built of a short peptide that adopts a critical structural motif in tauopathy fibrils found in Alzheimer’s Disease (AD) and Chronic Traumatic Encephalopathy (CTE). They serve as minimal prions with exquisite seeding competency, in vitro and in tau biosensor cells, for recruiting tau constructs ten times larger its size en route to AD or CTE fibril structures. We demonstrate that the generation of AD and CTE-like fibril structures is dramatically catalyzed in the presence of mini-AD prions and further influenced by salt composition in solution. Double Electron-Electron Resonance studies confirmed the preservation of AD-like folds across multi-generational seeding. Fibrils formed with the full AD/CTE-like core show strong seeding competency, with their templating effect dominating over the choice of salt composition that tunes the initial selection of AD- and CTE-like fibril populations. The mini-AD prions serve as a potent catalyst with templating capabilities that offer a novel strategy to design pathological tau fibril models.
Hydraulic Activation of the AsLOV2 photoreceptor
How proteins transduce environmental signals into mechanical motion remains a central question in biology. This study tests the hypothesis that blue light activation of AsLOV2 gives rise to concerted water movement that induce protein conformational extensions. Using electron and nuclear magnetic resonance spectroscopy, along with atomistic molecular dynamics simulations at high pressure, we find that activation, whether initiated by blue light or high pressure, is accompanied by selective expulsion of low-entropy, tetrahedrally coordinated “wrap” water from hydrophobic regions of the protein. These findings suggest that interfacial water serves as functional constituents to help reshape the protein’s free energy landscape during activation. Our study highlights hydration water as an active medium with the capacity to drive long-range conformational changes underlying protein mechanics and offers a new conceptual understanding for engineering externally controllable protein actuators for biomedical studies to smart materials.
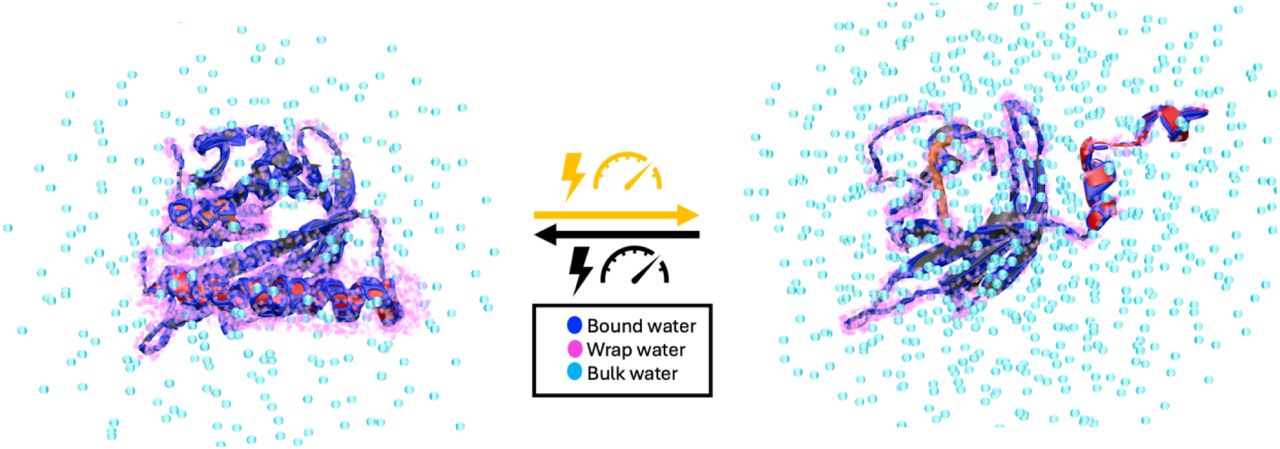
Hydraulic Activation of the AsLOV2 photoreceptor
How proteins transduce environmental signals into mechanical motion remains a central question in biology. This study tests the hypothesis that blue light activation of AsLOV2 gives rise to concerted water movement that induce protein conformational extensions. Using electron and nuclear magnetic resonance spectroscopy, along with atomistic molecular dynamics simulations at high pressure, we find that activation, whether initiated by blue light or high pressure, is accompanied by selective expulsion of low-entropy, tetrahedrally coordinated “wrap” water from hydrophobic regions of the protein. These findings suggest that interfacial water serves as functional constituents to help reshape the protein’s free energy landscape during activation. Our study highlights hydration water as an active medium with the capacity to drive long-range conformational changes underlying protein mechanics and offers a new conceptual understanding for engineering externally controllable protein actuators for biomedical studies to smart materials.

Water-directed pinning is key to tau prion formation
Featured in Northwestern Now, Phys.org, & Le Monde.
Tau forms fibrillar aggregates that are pathological hallmarks of a family of neurodegenerative diseases known as tauopathies. The synthetic replication of disease-specific fibril structures is a critical gap for developing diagnostic and therapeutic tools. This study debuts a strategy of identifying a critical and minimal folding motif in fibrils characteristic of tauopathies and generating seeding-competent fibrils from the isolated tau peptides. The 19-residue jR2R3 peptide (295 to 313) which spans the R2/R3 splice junction of tau, and includes the P301L mutation, is one such peptide that forms prion-competent fibrils. This tau fragment contains the hydrophobic VQIVYK hexapeptide that is part of the core of all known pathological tau fibril structures and an intramolecular counterstrand that stabilizes the strand–loop–strand (SLS) motif observed in 4R tauopathy fibrils. This study shows that P301L exhibits a duality of effects: it lowers the barrier for the peptide to adopt aggregation-prone conformations and enhances the local structuring of water around the mutation site to facilitate site-directed pinning and dewetting around sites 300-301 to achieve in-register stacking of tau to cross β-sheets. We solved a 3 Å cryo-EM structure of jR2R3-P301L fibrils in which each protofilament layer contains two jR2R3-P301L copies, of which one adopts a SLS fold found in 4R tauopathies and the other wraps around the SLS fold to stabilize it, reminiscent of the three- and fourfold structures observed in 4R tauopathies. These jR2R3-P301L fibrils are competent to template full-length 4R tau in a prion-like manner.
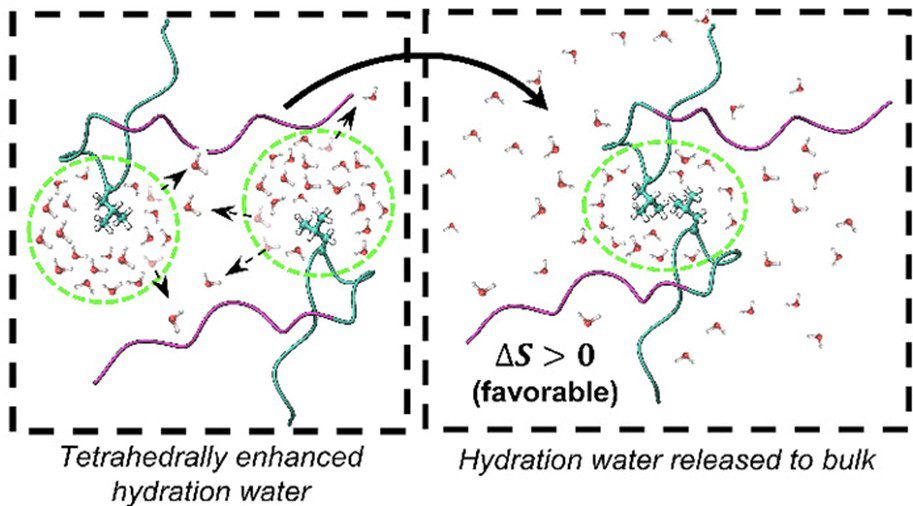
Water-directed pinning is key to tau prion formation
Featured in Northwestern Now, Phys.org, & Le Monde.
Tau forms fibrillar aggregates that are pathological hallmarks of a family of neurodegenerative diseases known as tauopathies. The synthetic replication of disease-specific fibril structures is a critical gap for developing diagnostic and therapeutic tools. This study debuts a strategy of identifying a critical and minimal folding motif in fibrils characteristic of tauopathies and generating seeding-competent fibrils from the isolated tau peptides. The 19-residue jR2R3 peptide (295 to 313) which spans the R2/R3 splice junction of tau, and includes the P301L mutation, is one such peptide that forms prion-competent fibrils. This tau fragment contains the hydrophobic VQIVYK hexapeptide that is part of the core of all known pathological tau fibril structures and an intramolecular counterstrand that stabilizes the strand–loop–strand (SLS) motif observed in 4R tauopathy fibrils. This study shows that P301L exhibits a duality of effects: it lowers the barrier for the peptide to adopt aggregation-prone conformations and enhances the local structuring of water around the mutation site to facilitate site-directed pinning and dewetting around sites 300-301 to achieve in-register stacking of tau to cross β-sheets. We solved a 3 Å cryo-EM structure of jR2R3-P301L fibrils in which each protofilament layer contains two jR2R3-P301L copies, of which one adopts a SLS fold found in 4R tauopathies and the other wraps around the SLS fold to stabilize it, reminiscent of the three- and fourfold structures observed in 4R tauopathies. These jR2R3-P301L fibrils are competent to template full-length 4R tau in a prion-like manner.

Hydrolytically Stable Phosphonate‐Based Metal–Organic Frameworks for Harvesting Water from Low Humidity Air
Harvesting water from air offers a promising solution to the global water crisis. However, existing sorbents often struggle in arid climates due to limitations such as low sorption capacities, hydrolytic instability, slow mass transport, high desorption enthalpy, and costly operation. Phosphonate-based metal–organic frameworks (MOFs), known for their exceptional water stability, have not been extensively explored for water harvesting. This study systematically investigates the performance of STA-12 (M═Co, Ni, Mg) and STA-16 (M═Co, Ni), a series of stable phosphonate-based MOFs, as water sorbents. STA-12 MOFs demonstrate remarkable adsorption at ultra-low humidity (<10%), while STA-16(Co) exhibits a high water uptake capacity of 0.54 g g−1 at 10–50% relative humidity (RH) and 0.72 g g−1 at 34% RH. Molecular simulations and solid-state NMR identified liquid-like water, critical for harvesting applications, as the key contributor to the superior sorption performance of STA-16(Co). Scalable aqueous synthesis methods are developed, producing tens of grams of MOFs per batch without high-pressure equipment. A prototype device incorporating STA-12(Ni) demonstrated the feasibility of these materials for real-world water harvesting, showcasing their potential to address water scarcity in arid regions
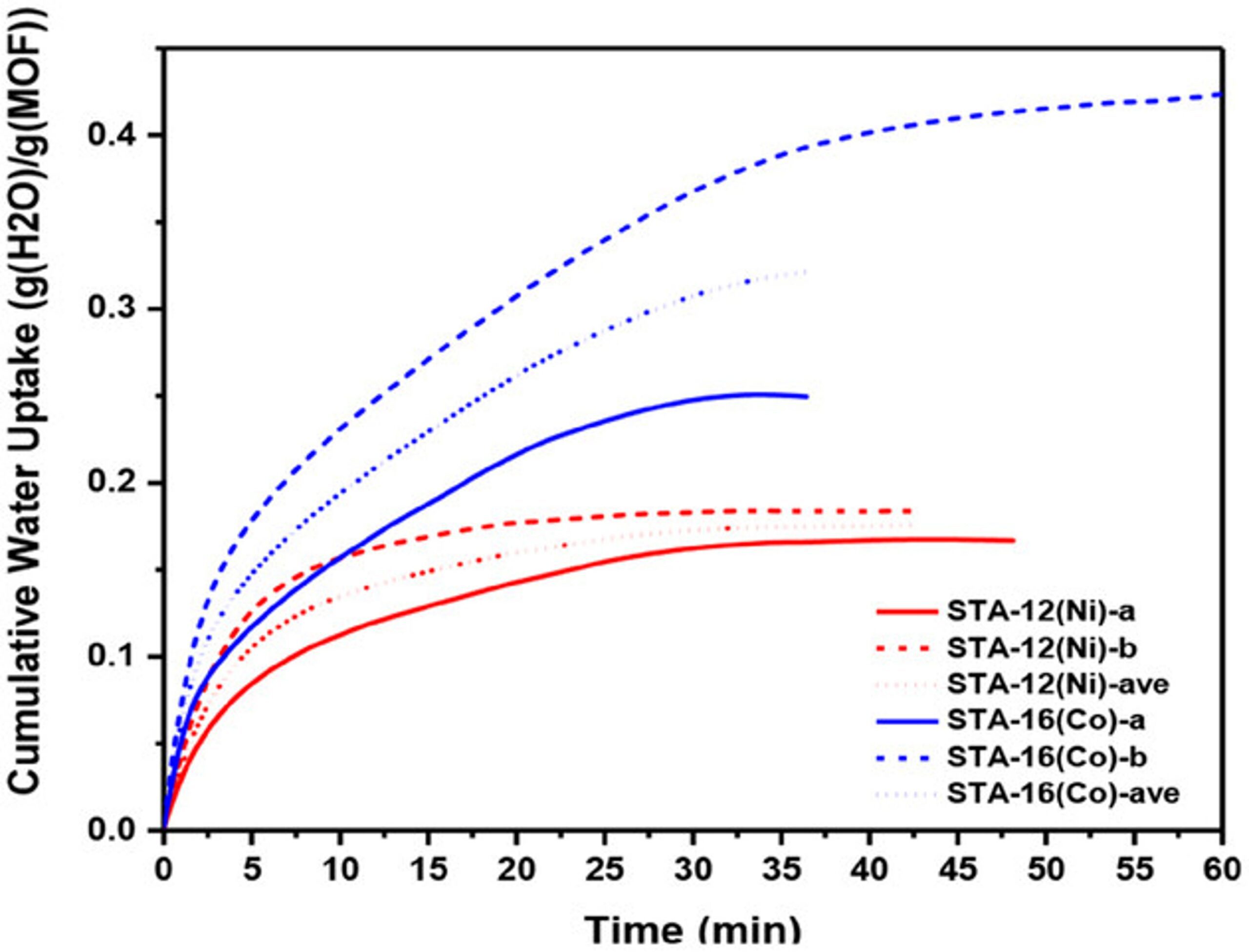
Hydrolytically Stable Phosphonate‐Based Metal–Organic Frameworks for Harvesting Water from Low Humidity Air
Harvesting water from air offers a promising solution to the global water crisis. However, existing sorbents often struggle in arid climates due to limitations such as low sorption capacities, hydrolytic instability, slow mass transport, high desorption enthalpy, and costly operation. Phosphonate-based metal–organic frameworks (MOFs), known for their exceptional water stability, have not been extensively explored for water harvesting. This study systematically investigates the performance of STA-12 (M═Co, Ni, Mg) and STA-16 (M═Co, Ni), a series of stable phosphonate-based MOFs, as water sorbents. STA-12 MOFs demonstrate remarkable adsorption at ultra-low humidity (<10%), while STA-16(Co) exhibits a high water uptake capacity of 0.54 g g−1 at 10–50% relative humidity (RH) and 0.72 g g−1 at 34% RH. Molecular simulations and solid-state NMR identified liquid-like water, critical for harvesting applications, as the key contributor to the superior sorption performance of STA-16(Co). Scalable aqueous synthesis methods are developed, producing tens of grams of MOFs per batch without high-pressure equipment. A prototype device incorporating STA-12(Ni) demonstrated the feasibility of these materials for real-world water harvesting, showcasing their potential to address water scarcity in arid regions

Passaging Human Tauopathy Patient Samples in Cells Generates Heterogeneous Fibrils with a Subpopulation Adopting Disease Folds
The discovery by cryo-electron microscopy (cryo-EM) that the neu-ropathological hallmarks of different tauopathies, including Alzheimer’s disease, corticobasal degeneration (CBD), and progressive supranuclear palsy (PSP), are caused by unique misfolded conformations of the protein tau is among the most profound developments in neurodegenerative disease research. To capitalize on these discoveries for therapeutic development, one must achieve in vitroreplication of tau fibrils that adopt the representative tauopathy disease folds, which represents a grand challenge for the field. A widely used approach has been seeded propagation using pathological tau fibrils derived from post-mortem patient samples in biosensor cells that expresses a fragment of the tau protein known as K18, or Tau4RD, containing the microtubule-binding repeat domain of tau as the substrate. The new insights from cryo-EM raised the question of whether the Tau4RD fragment is capable of adopting characteristic tau folds found in CBD and PSP patient fibrils, and whether cell-passaged and amplified tau fibrils can be used as seeds to achieve cell-free assembly of recombinant 4R tau into fibrils without the addition of cofactors. Using Double Electron Electron Resonance (DEER) spectroscopy, we discovered that cell-passaged pathological seeds generate heterogeneous fibrils that are, however, distinct between the CBD and PSP lysate-seeded fibrils, and vastly different from heparin-induced tau fibril structures. Moreover, the lysate-seeded fibrils contain a characteristic sub-population that resembles the disease fold corresponding to the respective starting patient sample. These findings indicate that templated propagation using CBD and PSP patient-derived fibrils is possible with a tau fragment that does not contain the entire pathological fibril core, but also that additional mechanisms must be tuned to converge on a homogeneous fibril population.

Passaging Human Tauopathy Patient Samples in Cells Generates Heterogeneous Fibrils with a Subpopulation Adopting Disease Folds
The discovery by cryo-electron microscopy (cryo-EM) that the neu-ropathological hallmarks of different tauopathies, including Alzheimer’s disease, corticobasal degeneration (CBD), and progressive supranuclear palsy (PSP), are caused by unique misfolded conformations of the protein tau is among the most profound developments in neurodegenerative disease research. To capitalize on these discoveries for therapeutic development, one must achieve in vitroreplication of tau fibrils that adopt the representative tauopathy disease folds, which represents a grand challenge for the field. A widely used approach has been seeded propagation using pathological tau fibrils derived from post-mortem patient samples in biosensor cells that expresses a fragment of the tau protein known as K18, or Tau4RD, containing the microtubule-binding repeat domain of tau as the substrate. The new insights from cryo-EM raised the question of whether the Tau4RD fragment is capable of adopting characteristic tau folds found in CBD and PSP patient fibrils, and whether cell-passaged and amplified tau fibrils can be used as seeds to achieve cell-free assembly of recombinant 4R tau into fibrils without the addition of cofactors. Using Double Electron Electron Resonance (DEER) spectroscopy, we discovered that cell-passaged pathological seeds generate heterogeneous fibrils that are, however, distinct between the CBD and PSP lysate-seeded fibrils, and vastly different from heparin-induced tau fibril structures. Moreover, the lysate-seeded fibrils contain a characteristic sub-population that resembles the disease fold corresponding to the respective starting patient sample. These findings indicate that templated propagation using CBD and PSP patient-derived fibrils is possible with a tau fragment that does not contain the entire pathological fibril core, but also that additional mechanisms must be tuned to converge on a homogeneous fibril population.

Localized Reconstruction of Multimodal Distance Distribution from DEER Data of Biopolymers
Pulsed Dipolar ESR Spectroscopy (PDS) is a uniquely powerful technique to characterize the structural property of intrinsically disordered proteins (IDPs) and polymers and the conformational evolution of IDPs and polymers, e.g. during assembly, by offering the probability distribution of segment end-to-end distances. However, it is challenging to determine distance distribution P(r) of IDPs by PDS because of the uncertain and broad shape information that is intrinsic to the distance distribution of IDPs. We demonstrate here that the Srivastava-Freed Singular Value Decomposition (SF-SVD) point-wise mathematical inversion method along with wavelet denoising (WavPDS) can aid in obtaining reliable shapes for the distance distribution, P(r), for IDPs. We show that broad regions of P(r) as well as mixed narrow and broad features within the captured distance distribution range can be effectively resolved and differentiated without a priori knowledge. The advantage of SF-SVD and WavPDS is that the methods are transparent, requiring no adjustable parameters, the processing of the magnitude for the probability distribution is performed separately for each distance increment, and the outcome of the analysis is independent of the user’s judgement. We demonstrate the performance and present the application of WavPDS and SF-SVD on model ruler molecules, model polyethylene glycol polymers with end-to-end spin labeling, and IDPs with pairwise labeling spanning different segments of the protein tau to generate the transparent solutions to the P(r)’s including their uncertainties and error analysis.

Localized Reconstruction of Multimodal Distance Distribution from DEER Data of Biopolymers
Pulsed Dipolar ESR Spectroscopy (PDS) is a uniquely powerful technique to characterize the structural property of intrinsically disordered proteins (IDPs) and polymers and the conformational evolution of IDPs and polymers, e.g. during assembly, by offering the probability distribution of segment end-to-end distances. However, it is challenging to determine distance distribution P(r) of IDPs by PDS because of the uncertain and broad shape information that is intrinsic to the distance distribution of IDPs. We demonstrate here that the Srivastava-Freed Singular Value Decomposition (SF-SVD) point-wise mathematical inversion method along with wavelet denoising (WavPDS) can aid in obtaining reliable shapes for the distance distribution, P(r), for IDPs. We show that broad regions of P(r) as well as mixed narrow and broad features within the captured distance distribution range can be effectively resolved and differentiated without a priori knowledge. The advantage of SF-SVD and WavPDS is that the methods are transparent, requiring no adjustable parameters, the processing of the magnitude for the probability distribution is performed separately for each distance increment, and the outcome of the analysis is independent of the user’s judgement. We demonstrate the performance and present the application of WavPDS and SF-SVD on model ruler molecules, model polyethylene glycol polymers with end-to-end spin labeling, and IDPs with pairwise labeling spanning different segments of the protein tau to generate the transparent solutions to the P(r)’s including their uncertainties and error analysis.

Phosphoryl group wires stabilize pathological tau fibrils as revealed by multiple quantum spin counting NMR
Hyperphosphorylation of the protein tau is one of the biomarkers of neurodegenerative diseases in the category of tauopathies. However, the molecular level, mechanistic, role of this common post- translational modification (PTM) in enhancing or reducing the aggregation propensity of tau is unclear, especially considering that combinatorial phosphorylation of multiple sites can have complex, non-additive, effects on tau protein aggregation. Since tau proteins stack in register and parallel to elongate into pathological fibrils, phosphoryl groups from adjacent tau strands with 4.8 Å separation must find an energetically favorable spatial arrangement. At first glance, this appears to be an unfavorable configuration due to the proximity of negative charges between phosphate groups from adjacent neighboring tau fibrils. However, this study tests a counterhypothesis that phosphoryl groups within the fibril core-forming segments favorably assemble into highly ordered, hydrogen-bonded, one-dimensionally extended wires under biologically relevant conditions. We selected two phosphorylation sites associated with neurodegeneration, serine 305 (S305p) and tyrosine 310 (Y310p), on a model tau peptide jR2R3-P301L (tau295-313) spanning the R2/R3 splice junction of tau, that readily aggregate into a fibril with characteristics of a seed-competent mini prion. Using multiple quantum spin counting (MQ-SC) by 31P solid-state NMR of phosphorylated jR2R3-P301L tau peptide fibrils, enhanced by dynamic nuclear polarization, we find that at least six phosphorous spins must neatly arrange in 1D within fibrils or in 2D within a protofibril to yield the experimentally observed MQ-coherence orders of four. We found that S305pstabilizes the tau fibrils and leads to more seeding-competent fibrils compared to jR2R3 P301L or Y310p. This study introduces a new concept that phosphorylation of residues within a core forming tau segment can mechanically facilitate fibril registry and stability due a hitherto unrecognized role of phosphoryl groups to form highly ordered, extended, 1D wires that stabilize pathological tau fibrils.
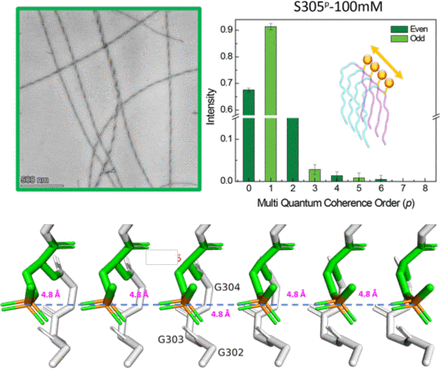
Phosphoryl group wires stabilize pathological tau fibrils as revealed by multiple quantum spin counting NMR
Hyperphosphorylation of the protein tau is one of the biomarkers of neurodegenerative diseases in the category of tauopathies. However, the molecular level, mechanistic, role of this common post- translational modification (PTM) in enhancing or reducing the aggregation propensity of tau is unclear, especially considering that combinatorial phosphorylation of multiple sites can have complex, non-additive, effects on tau protein aggregation. Since tau proteins stack in register and parallel to elongate into pathological fibrils, phosphoryl groups from adjacent tau strands with 4.8 Å separation must find an energetically favorable spatial arrangement. At first glance, this appears to be an unfavorable configuration due to the proximity of negative charges between phosphate groups from adjacent neighboring tau fibrils. However, this study tests a counterhypothesis that phosphoryl groups within the fibril core-forming segments favorably assemble into highly ordered, hydrogen-bonded, one-dimensionally extended wires under biologically relevant conditions. We selected two phosphorylation sites associated with neurodegeneration, serine 305 (S305p) and tyrosine 310 (Y310p), on a model tau peptide jR2R3-P301L (tau295-313) spanning the R2/R3 splice junction of tau, that readily aggregate into a fibril with characteristics of a seed-competent mini prion. Using multiple quantum spin counting (MQ-SC) by 31P solid-state NMR of phosphorylated jR2R3-P301L tau peptide fibrils, enhanced by dynamic nuclear polarization, we find that at least six phosphorous spins must neatly arrange in 1D within fibrils or in 2D within a protofibril to yield the experimentally observed MQ-coherence orders of four. We found that S305pstabilizes the tau fibrils and leads to more seeding-competent fibrils compared to jR2R3 P301L or Y310p. This study introduces a new concept that phosphorylation of residues within a core forming tau segment can mechanically facilitate fibril registry and stability due a hitherto unrecognized role of phosphoryl groups to form highly ordered, extended, 1D wires that stabilize pathological tau fibrils.

